|
||||||
| Off-Topic Lounge [WARNING: NO POLITICS] For all off-topic discussion topics. |
| View Poll Results: Will AI, humanoid robots or machines likely replace your job in your lifetime? | |||
| I'm too young to work, or I'm retired, so this poll doesn't apply to me |
|
3 | 9.38% |
| I've already been replaced. I'm broke and unemployed. |
|
1 | 3.13% |
| It will happen soon, or I'll likely retire before it does. |
|
2 | 6.25% |
| It won't happen soon, but it might happen in my lifetime. |
|
10 | 31.25% |
| It won't happen soon or in my lifetime, but it is inevitable. |
|
6 | 18.75% |
| Nope. Never. What I do is just too special, even in a million years. |
|
10 | 31.25% |
| Voters: 32. You may not vote on this poll | |||
 |
|
|
Thread Tools | Search this Thread |
|
|
#127 | |
|
Senior Member
Join Date: Mar 2017
Drives: Q5 + BRZ + M796
Location: Santa Rosa, CA
Posts: 7,884
Thanks: 5,668
Thanked 5,810 Times in 3,300 Posts
Mentioned: 70 Post(s)
|
Quote:
So despite the claims, they have approved human trials??? Maybe the complaints weren't as bad as the article is suggesting.
__________________
My Build | K24 Turbo Swap | *K24T BRZ SOLD*
|
|
|
|

|
| The Following User Says Thank You to Irace86.2.0 For This Useful Post: | Ohio Enthusiast (09-26-2023) |
|
|
#128 |
|
Senior Member
Join Date: May 2011
Drives: '23 BRZ
Location: Providence, RI
Posts: 4,672
Thanks: 1,439
Thanked 4,012 Times in 2,098 Posts
Mentioned: 85 Post(s)
|
|
|
|

|
| The Following 2 Users Say Thank You to ZDan For This Useful Post: | Dadhawk (09-26-2023), soundman98 (09-26-2023) |
|
|
#129 |
|
The Dictater
Join Date: Apr 2017
Drives: '13 Red Scion FRS
Location: MD, USA
Posts: 9,683
Thanks: 26,748
Thanked 12,739 Times in 6,313 Posts
Mentioned: 88 Post(s)
|
All kinds of animals are bred as test subjects, and that's just how testing works.
That being said, Musk making false statements to hide his company's shortcomings such that the investment stream continues to flow is not surprising at all. This is a common theme and why I personally want nothing to do with him or any companies he runs.
__________________
If a picture is worth a thousand words, a model is worth ten thousand pictures.
Also: "Build Thread" |
|
|

|
| The Following 2 Users Say Thank You to Spuds For This Useful Post: | blsfrs (09-27-2023), x808drifter (09-30-2023) |
|
|
#130 | |||
|
Senior Member
Join Date: Mar 2017
Drives: Q5 + BRZ + M796
Location: Santa Rosa, CA
Posts: 7,884
Thanks: 5,668
Thanked 5,810 Times in 3,300 Posts
Mentioned: 70 Post(s)
|
Quote:
Quote:
Quote:
Yes, Musk is going to conceal and placate. That is what all CEOs do. This isn't a surprise to me, but some of these charges seem hilarious: They are claiming they are going to put a safe device on the market, and thats why you should invest." Well duh! That is the plan. It isn't to put an unsafe device on the market, so people should invest. It wasn't just animal testing. It was development of a product and procedure for implanting the device. Human trials will probably require further development, but that will be more like testing and fine tuning. The candidates are willful participants who are going to be dead in years or are quads. Investors don't care about ten monkeys, even if that was half of the subjects. They care about whether Musk has the ability to form a team capable of making the product reality. The fact that this is at human trials from animal trials suggests the claims made in the article weren't significant enough. 90% of pharmaceutics fail during the drug development process (Source), and this is true for medical devices too (Source), so moving a product to human trials in spite of these claims makes me skeptical of the claims of gross negligence.
__________________
My Build | K24 Turbo Swap | *K24T BRZ SOLD*
|
|||
|
|

|
| The Following User Says Thank You to Irace86.2.0 For This Useful Post: | blsfrs (09-27-2023) |
|
|
#131 | |
|
The Dictater
Join Date: Apr 2017
Drives: '13 Red Scion FRS
Location: MD, USA
Posts: 9,683
Thanks: 26,748
Thanked 12,739 Times in 6,313 Posts
Mentioned: 88 Post(s)
|
Quote:
Who do you suppose makes the decision to move a product to human trials?
__________________
If a picture is worth a thousand words, a model is worth ten thousand pictures.
Also: "Build Thread" |
|
|
|

|
| The Following User Says Thank You to Spuds For This Useful Post: | blsfrs (09-27-2023) |
|
|
#132 | |
|
Senior Member
Join Date: Mar 2017
Drives: Q5 + BRZ + M796
Location: Santa Rosa, CA
Posts: 7,884
Thanks: 5,668
Thanked 5,810 Times in 3,300 Posts
Mentioned: 70 Post(s)
|
Quote:
Looks like here is another timeline for FSD release for FSD 12.0 supposedly released this year. Maybe this one will be it: https://www.carexpert.com.au/car-new...ting-from-beta Like the FAA has just finished their investigation with SpaceX and is demanding actions before being cleared for another launch, the FDA does the approvals for trials, and they denied Neuralink's initial application, citing the need for changes before Neuralink would be cleared for human trials. Apparently that happened. https://www.reuters.com/investigates...link-musk-fda/
__________________
My Build | K24 Turbo Swap | *K24T BRZ SOLD*
|
|
|
|

|
| The Following User Says Thank You to Irace86.2.0 For This Useful Post: | blsfrs (09-27-2023) |
|
|
#133 | ||
|
The Dictater
Join Date: Apr 2017
Drives: '13 Red Scion FRS
Location: MD, USA
Posts: 9,683
Thanks: 26,748
Thanked 12,739 Times in 6,313 Posts
Mentioned: 88 Post(s)
|
Quote:
https://www.fda.gov/regulatory-infor...edical-devices https://www.fda.gov/patients/learn-a...opment-process Edit, I must have missed the IND section of clinical trials. Still, they note that an actual hold is rare and more often there are recommendations made to improve safety. Quote:
__________________
If a picture is worth a thousand words, a model is worth ten thousand pictures.
Also: "Build Thread" Last edited by Spuds; 09-27-2023 at 09:34 AM. |
||
|
|

|
| The Following User Says Thank You to Spuds For This Useful Post: | Irace86.2.0 (09-27-2023) |
|
|
#134 | |
|
Senior Member
Join Date: Mar 2017
Drives: Q5 + BRZ + M796
Location: Santa Rosa, CA
Posts: 7,884
Thanks: 5,668
Thanked 5,810 Times in 3,300 Posts
Mentioned: 70 Post(s)
|
Quote:
__________________
My Build | K24 Turbo Swap | *K24T BRZ SOLD*
|
|
|
|

|
|
|
#135 |
|
The Dictater
Join Date: Apr 2017
Drives: '13 Red Scion FRS
Location: MD, USA
Posts: 9,683
Thanks: 26,748
Thanked 12,739 Times in 6,313 Posts
Mentioned: 88 Post(s)
|
I believe that was referring to premarket approvals after clinical trials, not the investigational new drug approval before clinical trials. I'm referring to the latter.
__________________
If a picture is worth a thousand words, a model is worth ten thousand pictures.
Also: "Build Thread" |
|
|

|
| The Following User Says Thank You to Spuds For This Useful Post: | MyHybridBurnsGasAndTires (09-28-2023) |
|
|
#136 | |
|
Senior Member
Join Date: Mar 2017
Drives: Q5 + BRZ + M796
Location: Santa Rosa, CA
Posts: 7,884
Thanks: 5,668
Thanked 5,810 Times in 3,300 Posts
Mentioned: 70 Post(s)
|
Quote:
The FDA is involved in every stage from preclinical (animal) studies to the final approval to post-clinical studies to pull something off the market. You mention that holds and denials are rare, but I hope you aren't suggesting that the FDA is soft with allowing products to move through the stages. Holds and denials are rare because companies invest millions and do everything they can to be in compliance and to make a product that will be successful through the whole process. Tying this back to Neuralink, and in light of them being denied once by the FDA, the fact that they are cleared for Phase I (human) trials with a Class III device suggests they have done their due diligence with pretrials and showed enough efficacy and safety to move forward, given the rigorous demands of the FDA. https://www.fda.gov/media/124862/download https://www.fda.gov/drugs/informatio...cess-continued 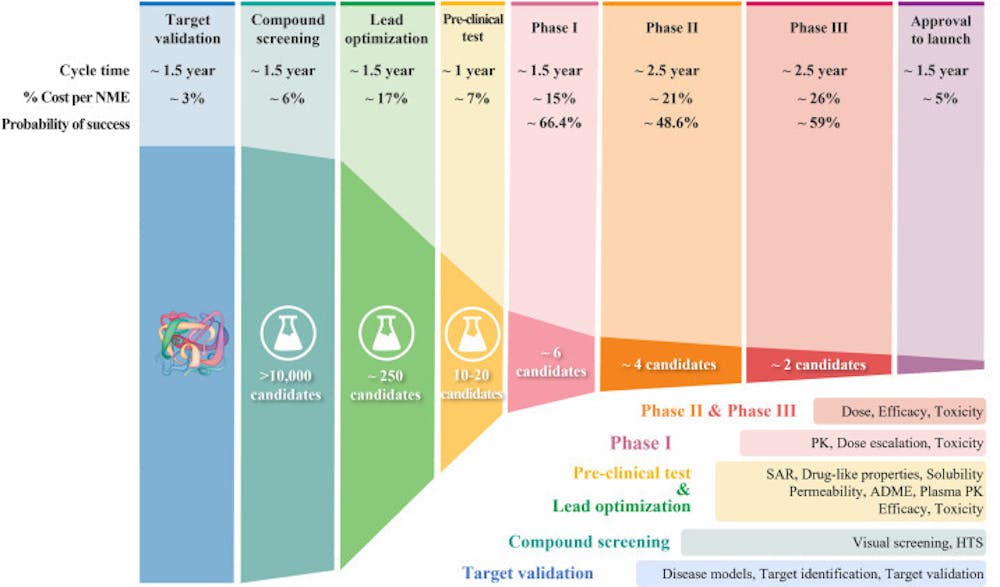 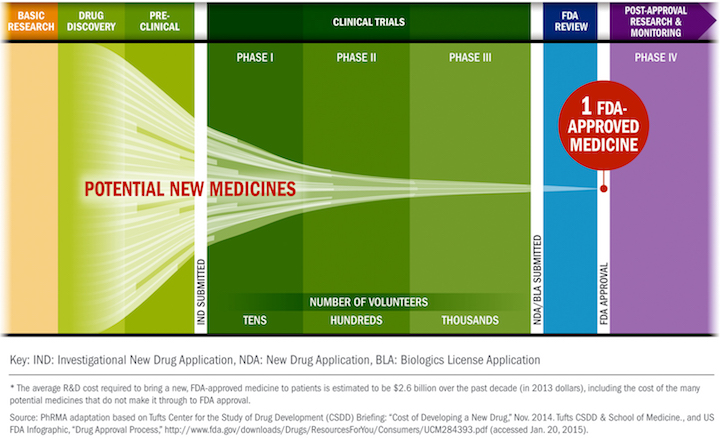
__________________
My Build | K24 Turbo Swap | *K24T BRZ SOLD*
|
|
|
|

|
|
|
#137 | |
|
The Dictater
Join Date: Apr 2017
Drives: '13 Red Scion FRS
Location: MD, USA
Posts: 9,683
Thanks: 26,748
Thanked 12,739 Times in 6,313 Posts
Mentioned: 88 Post(s)
|
Quote:
The second link notes the IND existence, but is more focused on the NDA, which occurs after clinical trials. I still haven't seen any indication that the FDA is involved prior to the IND. The source I provided from the FDA itself described the IND process and specifically said holds are rare, usually because biotech firms do their due diligence. The fact that there was already a hold on Neuralink's IND (or device equivalent) is a pretty big red flag in that context, indicating that the testing and process was not up to the FDA standard the first time around. None of the charts you provided indicate the FDA actually has any involvement in preclinical trials. My point is that it is up to the company to QA their own process, and for the most part self regulate. The first time the FDA has to be involved is at the IND, and what they are looking for is whether the company's report indicates that the risks are understood, and that appropriate measures have been taken in the process of understanding those risks. It's self-reported. That's why empowered QA is important. Again, Musk has shown multiple times he is willing to omit pertinent information, or even outright lie, to make some product appear more successful than it really is. I would guess that means he is also willing to pressure his employees to do so.
__________________
If a picture is worth a thousand words, a model is worth ten thousand pictures.
Also: "Build Thread" |
|
|
|

|
|
|
#138 | |
|
Senior Member
Join Date: Mar 2017
Drives: Q5 + BRZ + M796
Location: Santa Rosa, CA
Posts: 7,884
Thanks: 5,668
Thanked 5,810 Times in 3,300 Posts
Mentioned: 70 Post(s)
|
Quote:
https://www.fda.gov/cosmetics/produc...tory%20Animals. https://www.nal.usda.gov/animal-heal...0Animal%20Care. Part of pretrial animal studies is to find a therapeutic range and then to find a toxic range. Sometimes studies don't go further to human trials because the animals develop tumors or die, even if it is at super therapeutic doses, sometimes thousands of times higher. Everything that happens in pretrials is reportable on applications for human trials. Failing to do efficacy studies can be grounds for a denial to move to human trials, so compliance and due diligence is a must. If there are reports of negligence, safety concerns, rights violations, etc, you bet the government will show up like they will for a restaurant, and you bet the results of pretrials and adherence to guidelines play in their decision to approve or deny applications, but this tends to not be an issue, as companies invest in products and want to be compliant in order to get approved. https://www.fda.gov/media/93963/download Musk and company could lie, but this would be bad--very bad. There is a reason they were denied the IND, and it was because they likely WERE transparent and didn't lie. Being denied is less important than the fact that they were approved, which means they made the necessary changes or provided the necessary data to meet safety and efficacy guidelines. Also, what do you think will happen in Phase I human trials if Neuralink is as cavalier as you suggest? They will get shut down hard and fast. Lots of money down the tube. Loss of confidence with investors. They are going to do everything they can to succeed. https://www.fda.gov/medical-devices/...ory-misconduct
__________________
My Build | K24 Turbo Swap | *K24T BRZ SOLD*
|
|
|
|

|
|
|
#139 | |
|
Senior Member
Join Date: May 2011
Drives: '23 BRZ
Location: Providence, RI
Posts: 4,672
Thanks: 1,439
Thanked 4,012 Times in 2,098 Posts
Mentioned: 85 Post(s)
|
Quote:
I'm not against capitalism, but *unregulated* capitalism is actively eating itself. It rewards psychopaths like Musk who have zero qualms about exploiting their employees and their customers as much as possible while neglecting regulations. F that guy, he's the worst... |
|
|
|

|
| The Following User Says Thank You to ZDan For This Useful Post: | soundman98 (09-30-2023) |
|
|
#140 | |
|
Senior Member
Join Date: Mar 2017
Drives: Q5 + BRZ + M796
Location: Santa Rosa, CA
Posts: 7,884
Thanks: 5,668
Thanked 5,810 Times in 3,300 Posts
Mentioned: 70 Post(s)
|
Quote:
I don't think Neuralink is cheating in any way similar to what VW did, nor has Musk, for the most part. He definitely is guilty of union busting. He was guilty of trying to manipulate the stock market to screw over short sellers. I think he placates and smoothes, as any CEO/politician would. He minimizes the problems and embellishes the successes, although he is also fine talking about the problems and hell, trials and tribulations that his companies or himself has gone through, whether we are talking about Tesla, SpaceX, SolarCity, X, etc. Remember, there are hundreds of thousands of people begging for this technology. I could list all the conditions, but I don't know if that is necessary to drive home the point that this type of technology is profound. Musk will have no problem finding volunteers. They already are reaching out to him on X. I don't believe he will be taking short cuts with people's lives during clinical trials. He will get shut down hard. Check it out.
__________________
My Build | K24 Turbo Swap | *K24T BRZ SOLD*
|
|
|
|

|
|
|